 
Large-vessel endothelial cells possess Weibel-Palade bodies, whereas microvascular endothelial cells do not contain this organelle ( 8). Although the exact vessel size is an approximation, high airway pressure closes blood vessels smaller than approximately 25 μm in diameter (so-called “alveolar vessels”) and opens blood vessels larger than approximately 25 μm (so-called “extraalveolar vessels”) ( 7). Physiologists recognize a functional border between the macro- and the microcirculations that is revealed by high airway pressure. However, the anatomy of pulmonary endothelial cells change as large blood vessels decrease in size, and pulmonary endothelial cells ultimately become capillaries ( 5, 6). These cells are closely apposed to their neighbors and reside on a clearly delineated basement membrane. In recent years, a growing appreciation for the heterogeneity of endothelium within each of these three classifications, but most prominently within the continuous subtype, has been realized.Ĭontinuous type endothelial cells line blood vessels in the pulmonary circulation. Fenestrated endothelia can be contrasted further because they display 60 to 80 nm transcellular openings with thin diaphragms visible en face. Discontinuous endothelia can be contrasted with this description by their constitutive prominence of intercellular gaps and an absence of basement membrane. Most endothelium is of the continuous subtype, possessing close apposition among adjacent cells and an absence of fenestrations. In a broad sense, endothelium has been classified on this morphological basis as being continuous, discontinuous (e.g., sinusoidal), or fenestrated ( 1). 
Scientists and clinicians have recognized a morphological diversity in endothelial cells ( 1– 4). This ability of endothelium to rapidly adapt to environment pressures is a cardinal feature of vascular health. 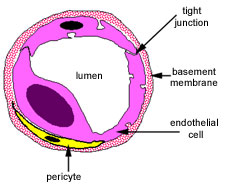
The endothelium dynamically adjusts its barrier integrity (thereby influencing water, solute, and macromolecular access to the interstitial compartment), provides a gateway for circulating cells to access tissue, produces vasoconstrictor and vasodilator substances, and affects coagulation and hemostasis ( 1). In this position, they interpret the physical (mechanical) and chemical environments of blood and tissue and, in turn, adjust their behavior to optimize homeostasis ( 1). Rather, development of novel therapeutics based upon anatomically restricted expression of molecular signatures may be developed to better combat vascular disease.Įndothelial cells are positioned between blood and the interstitial space. Although endothelium is an attractive therapeutic target in acute lung injury and other vascular disorders, the growing awareness of pulmonary endothelial cell heterogeneity increasingly suggests that a panendothelial cell approach is suboptimal. Whereas activation of the TRPV4 channel causes alveolar flooding, activation of the α1G T-type calcium channel promotes P-selectin surface translocation, events specific to the microcirculation. Pulmonary microvascular endothelial cells express calcium channels not found in extraalveolar endothelial cells, including the vanilloid family transient receptor potential 4 channel and the α1G T-type calcium channel. This cellular feature is observed in culture and in the intact microcirculation, prompting a reevaluation of the key physiological principles that control permeability and the fate of fluid (or exudate) once it leaves the circulation. One of the most striking features of pulmonary microvascular endothelial cells is that they possess a highly impermeable barrier with respect to pulmonary artery or vein endothelial cells. In recent years there has been an increasing appreciation of the functional heterogeneity that exists between extraalveolar and alveolar endothelial cells. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
